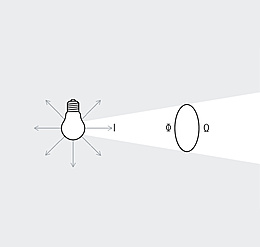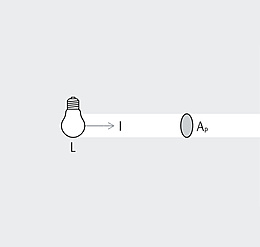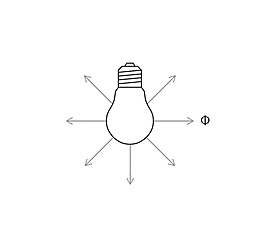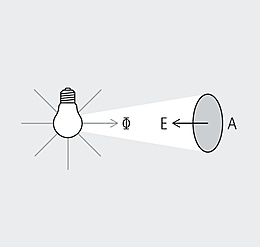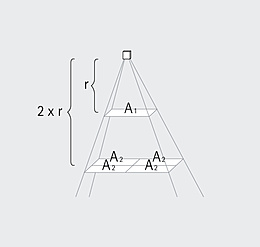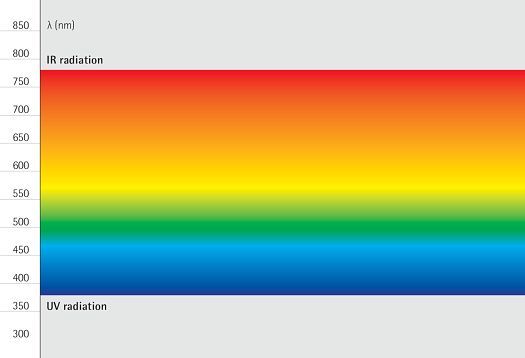
Spectrum is the term used to describe the distribution of electromagnetic waves. Only a small part is visible to humans in the range between 380 and 780 nanometers (nm) – this is what we call light. Colloquially, we talk about "visible light". The spectral distribution of a light source determines its light color and its color rendering. Depending on the method of generation of the light, basic types of spectra can be distinguished between: the continuous light spectrum (daylight, thermal radiators and white LEDs), the line spectrum (low pressure discharge lamps) and the band spectrum (high pressure discharge lamps).